10th Science Unit 3 Book Back Answers – Thermal Physics:
Samacheer Kalvi 10th Standard Science Book Back Questions with Answers PDF uploaded and the same given below. Class-tenth candidates and those preparing for TNPSC exams can check the New Science Book Back Question and Answers PDF below. Samacheer Kalvi Class 10th Std Science Book Back Answers Physics Unit 3 – Thermal Physics Solutions are available below. Complete solutions for all 23 Units’ book back questions with a solutions guide uploaded and available on our site. Check the complete Samacheer Kalvi 10th Science Unit 3 Answers below:
We also provide class 10th other units Science Book Back One and Two Mark Solutions Guide on our site. Students looking for a new syllabus 10th standard Science Physics Unit 3 Thermal Physics Book Back Questions with Answer PDF
For the complete Samacheer Kalvi 10th Science Book Back Solutions Guide PDF, check the link – Samacheer Kalvi 10th Science Book Back Answers
Samacheer Kalvi 10th Science Book Back Answers Physics Unit 3 – Thermal Physics
Samacheer Kalvi 10th Science Book Subject One Mark, Two Mark Guide questions and answers are below. Check Science Book Back Questions with Answers. Take the printout and use it for exam purposes.
For Samacheer Kalvi 10th Science Book PDF, check the link – 10th Science Book PDF
10th Science – Physics
Unit 3: Thermal Physics
I. Choose the best answer:
1. The value of the universal gas constant
a) 3.81 mol–1 K–1 b) 8.03 mol–1 K–1
c) 1.38 mol–1 K–1 d) 8.31 mol–1 K–1
Ans : d) 8.31 mol–1K–1
2. If a substance is heated or cooled, the change in mass of that substance is
a) positive b) negative c) zero d) none of the above
Ans: c) zero
3. If a substance is heated or cooled, the linear expansion occurs along the axis of
a) X or –X b) Y or –Y c) both (a) and (b) d) (a) or (b)
Ans : c) both (a) and (b)
4. Temperature is the average of the molecules of a substance.
a) difference in K.E and P.E b) sum of P.E and K.E
c) difference in T.Eand P.E d) difference in K.E and T.E
Ans: c) difference in T.E. and P.E
5. In the Given diagram, the possible direction of heat energy transformation
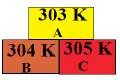
(a) A ← B, A ← C, B ← C
(b) A → B, A → C, B → C
(c) A → B, A ← C, B → C
(d) A ← B, A → C, B ← C.
Ans: a) A ← B, A ← C, B ← C
II. Fill in the blanks
1. The value of the Avogadro number. Ans: 6.023 × 1023/mol
2. The temperature and heat are quantities. Ans: Scalar
3. One calorie is the amount of heat energy required to raise the temperature of water through. Ans: 1g ; 1°C
4. According to Boyle’s law, the shape of the graph between pressure and reciprocal of volume is.
Ans: straight line
III. State whether the following statements are true or false, if false explain why?
1. For a given heat in liquid, the apparent expansion is more than that of real expansion.
Ans: False. For a given heat in liquid, the real expansion is more or less than that of apparent expansion.
2. Thermal energy always flows from a system at a higher temperature to a system at a lower temperature Ans: True.
3. According to Charles’s law, at constant pressure, the temperature is inversely proportional to volume.
Ans: False. According to Charles’s law, at constant pressure, the volume is directly proportional to temperature.
IV. Match the following:
| Column-I | Column-II |
| 1. Linear expansion | (a) change in volume |
| 2. Superficial expansion | (b) hot body to cold body |
| 3. Cubical expansion | (c) 1.381 × 10-23 JK-1 |
| 4. Heat transformation | (d) change in length |
| 5. Boltzmann constant | (e) change in an area |
Answer:
1. (d) change in length
2. (e) change in the area
3. (a) change in volume
4. (b) hot body to cold body
5. (c) 1.381 × 10-23 JK-1
V. Assertion and Reason:
a. Both the assertion and the reason are true and the reason is the correct explanation of the assertion.
b. Both the assertion and the reason are true but the reason is not the correct explanation of the assertion.
c. Assertion is true but the reason is false.
d. Assertion is false but the reason is true.
1. Assertion: There is no effects on other ends when one end of the rod is only heated.
Reason: Heat always flows from a region of lower temperature to a higher temperature of the rod.
Ans: c. Assertion is true but the reason is false.
2. Assertion: Gas is highly compressible than solid and liquid.
Reason: Interatomic or intermolecular distance in the gas is comparably high.
Ans: c. The assertion is true but the reason is false
Other Important links for 10th Science Book Answers Solutions:
For the 10th Standard Science Unit 4 book back question and answers solutions PDF, check the link – Samacheer kalvi 10th Science Unit 4 – Electricity
Click here for the complete Samacheer Kalvi 10th Science Book Back Solution Guide PDF – 10th Science Book Back Solutions
